Effects of treadmill training on motor function balance and spasticity reduction in children with cerebral palsy: A randomized clinical trail
DOI:
https://doi.org/10.52567/trehabj.v9i02.96Keywords:
balance, cerebral palsy, motor function, treadmill trainingAbstract
Background: Cerebral palsy (CP) is the leading cause of childhood motor disability, often associated with impaired, poor balance and spasticity, which limits daily activities and reduces quality of life. Treadmill training has emerged as a promising therapeutic intervention aimed at enhancing these impairments.
Objective: To evaluate the effects of treadmill training on motor function, balance, and Spasticity in Spastic CP children
Methods: The randomized control trial was conducted at Helping Hand Comprehensive Physical Rehab Centre, Lower Dir, from June 2024 to January 2025. A total of n=36 children with spastic CP aged 4-12 with GMFCS levels I and II were included in the study. The GMFCS, pediatric balance scale (PBS), and modified Ashworth scale (MAS) were used to assess motor function, balance, and spasticity. Out of n=36 CP children, n=18 individuals received treadmill training along with conventional therapy, and n=18 received conventional therapy only. The assessments were done at the beginning, after 4 weeks, 8 weeks, and 12 weeks of training.
Results: The mean age of 8.17±2.21 years, having 42.6% male and 24.15% female. A non-significant interaction effect between intervention and time effect on spasticity {F(3,102)=0.81, p=0.489, ηp²=0.02} measured by modified Ashworth scale. While in PBS {F(3,102)=36.41, p=0.489, ηp²=0.517} significant interaction effect observed. Regarding motor functions, the Friedman test did not indicate overall significant (p≥0.05) change in GMFCS scores over time in both the experimental and control groups, and Wilcoxon signed-rank tests also confirmed insignificant (p≥0.05) changes between adjacent time points.
Conclusion: Progressive treadmill protocol when combined with conventional therapy, can produce clinically meaningful improvements in balance and spasticity in children with CP, even though it may not alter gross motor function classification in the short term.
Clinical Trail #: NCT06463301
INTRODUCTION
Cerebral palsy (CP) is a non-progressive group of permanent movement disorders and the leading cause of childhood motor disability, often associated with impaired motor function, poor balance, and spasticity[1]. These impairments significantly limit a child's ability to perform daily activities and reduce their quality of life[2]. Cerebral palsy (CP) is attributed to factors such as birth asphyxia, maternal anemia, and pregnancy-induced hypertension[3]. The prevalence of Cerebral palsy affects approximately 1.7 million people globally. While with limited demographic data in Pakistan, A study in Karachi revealed a male-to-female ratio of 1.4:1, with prevalent cases of spastic tone (53.4%)[4].
There are several therapeutic approaches for treating spasticity in children with non-progressive chronic encephalopathy[5, 6]. Physiotherapy improves function, posture, balance, and gait in cerebral palsy and effective intervention requires careful assessment of impairments. The strategies should be focused on improving activity rather than solely reducing impairments[7, 8]. The combination of SPT and Partial Body Weight-Supported Treadmill Trainingrtial Body Weight-Supported Treadmill Training (PBWSTT improves gross motor function[9, 10].
Treadmill training has emerged as a promising therapeutic intervention aimed at enhancing motor function, improving balance, and reducing spasticity in children with CP[11]. The rhythmic, repetitive nature of treadmill walking stimulates neuroplasticity, encourages more normal gait patterns, and facilitates improvements in lower limb coordination and strength. Additionally, treadmill training can provide a safe, controlled environment to practice weight-bearing and stepping, which are critical for functional mobility[12]. While traditional physical therapy remains the cornerstone of CP management[13].
There is growing interest in treadmill training as a task-specific intervention, that can potentially offer greater functional improvements. Although previous studies have investigated treadmill training in children with cerebral palsy, most research has been limited by small sample sizes, and high-quality randomized clinical trial evidence specifically examining how training affects motor function, balance, and spasticity simultaneously. The current study to evaluate the effects of treadmill training on motor function, balance, and Spasticity in Spastic CP children
MATERIALS AND METHODS
Study Design: Participants: The approval obtained from the research and ethical committee of the Faculty of Rehabilitation and Allied Health Sciences Islamabad (RIPHAH/FR&AHS/letter-01826) and institutional head of Helping Hand Institute of Rehabilitation Sciences, in Lower Dir Talash from January to June 2024. The children diagnosed with cerebral palsy, aged between 04-12 years, having I or II levels of dependency on GMFCS with will cognitive abilities moreover the children misdiagnosis of CP, mental retardation, other neurological abnormalities, uncontrolled seizures, prior similar training, multiple contractures, significant respiratory issues, use of muscle relaxants, and hearing or communication problems were excluded from the study. A non-probability convenient sampling technique was used for sample collection.
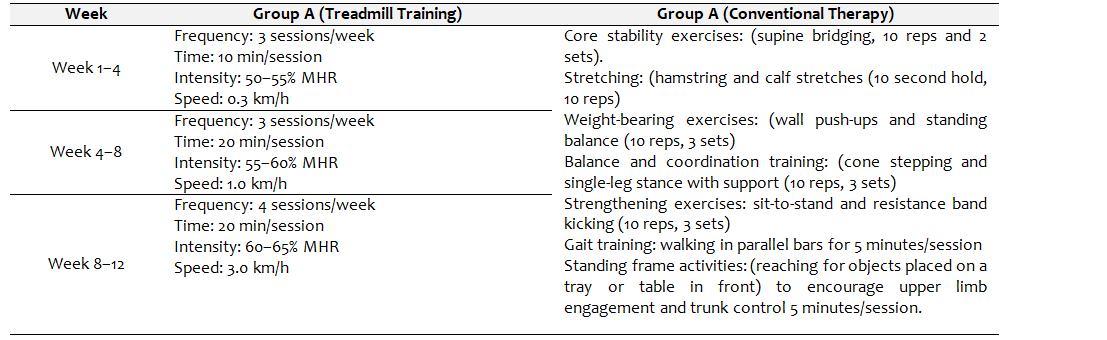
Interventions: The eligible participants were randomly allocated in a 1:1 ratio into the treadmill training group (group A, n=18) and the conventional physical therapy group (group B, n=18). The participants received both intervention over 12 weeks, and outcomes assessment was done at baseline, week 4, week 8, and week 12.
Group A (Treadmill Training+Conventional Therapy): Participants in the experimental group received treadmill training in addition to conventional physical therapy. The treadmill intervention was based on the FITT principle, with progressive increases in speed, duration, and intensity across the 12 weeks. As a supportive harness system was not available, two trained physical therapists were present during each session to ensure safety. One therapist stood behind the child and the other at the side, assisting in balance, posture correction, and fall prevention. Participants were instructed to treadmill's parallel bars for added support. A mirror placed in front of the treadmill provided visual feedback to encourage upright posture and correct gait mechanics. Verbal cues were given throughout the sessions to guide and motivate the children.
Group B (Conventional Therapy Only): Group B received conventional physical therapy only, which included core stability exercises, sustained stretching, Weight-bearing exercises for upper and lower limbs, Balance and coordination training, strengthening exercises for upper and lower limbs, Gait training using parallel bars and standing frame activities. All therapy sessions were supervised by a physiotherapist, with reassessments conducted at four-week intervals for both groups.
Table 1. Detailed Intervention Protocol
Outcome Measures: In addition to demographic data age, BMI, and level of GMFCS were obtained. The GMFCS classified children with cerebral palsy based on their self-initiated movement abilities. Levels range from I (most functional) to V (most severe limitation). It is a validated and reliable tool (Kappa>0.90) as a global indicator of gross motor functions. The PBS is a valid and reliable tool (ICC>0.95) used to measure static and dynamic balance through 14 functional tasks. Each item is scored from 0 to 4 with scores ranging from 0 to 56 and higher scores indicating better balance. The MAS is a reliable tool (ICC=0.61–0.91) that was used to assess muscle tone and grade the level of spasticity during passive stretching. The scores range from 0 (no increase in tone) to 4 (rigid in flexion or extension) and lower scores indicate a reduction in spasticity.
Sample Size: 36 samples were estimated using G power with a small effect size (0.2) and an α error margin of 0.05. To reduce β error probability, the power (1- β) was set to 0.95%. 46 children with CP were screened for inclusion criteria; 18 individuals did not meet the selection criteria and were therefore excluded from the study. A total of 36 participants were randomly assigned to groups A (n=18) and B (n=18) respectively. (Figure 1)
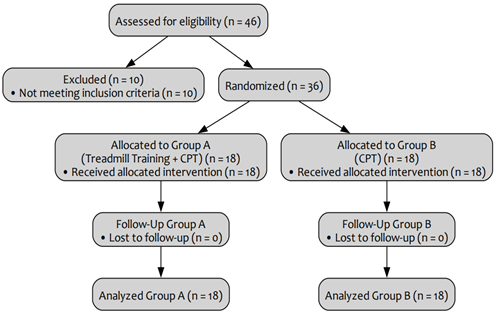
Figure 1: Consort Diagram
Randomization and blinding: Randomization was performed using the sealed enveloped method and a computerized random number generator. The random allocation was carried out by a biostatistician who was not involved in data collection. The random numbers were then written on index cards and sealed in a thick, opaque envelope before the investigation began. Following written informed consent, the physical therapist opened the envelope and administered the appropriate therapies to the patients. The assessing physical therapist was blinded to the intervention hence the trial was single-blind.
Statistical methods: The data analysis was done through the SPSS ver 26, and the significance level was set at p<0.05. A mixed-design ANOVA was applied to assess the interaction between Interventions and assessment time at baseline, 4th week, 8th week, and 12th week on motor function balance and spasticity. For the within main effects ANOVA with repeated measures and independent t-test for between the group comparison were applied.
RESULTS
The study sample comprised n=36 CP children ranging in age from 5 to 13 years, with a mean age of 8.17±2.21 years. In terms of Body Mass Index (BMI), values ranged from 18.29 to 51.22, with a mean BMI of 33.97±7.17 kg/m². A total of n=23(63.89%) males and the remaining n=13(36.11%) were female included in the study.
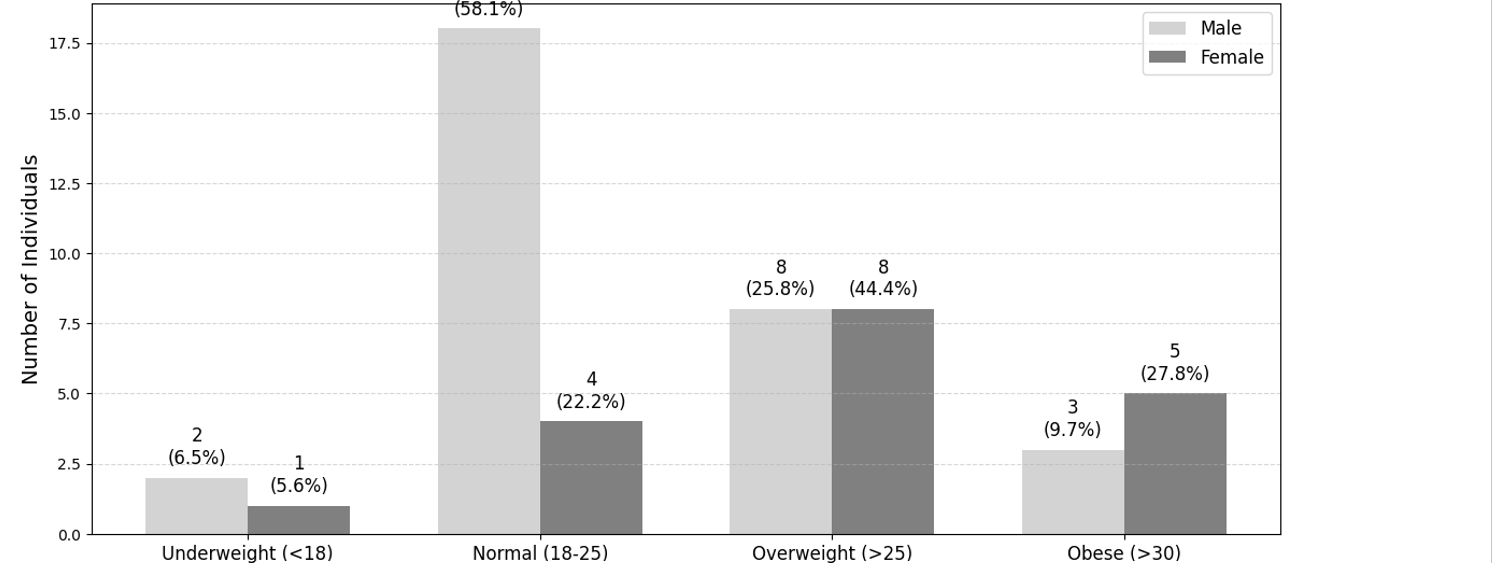
Figure 2: Gender based BMI distribution
The sphericity-assumed mixed ANOVA showed a non-significant interaction effect between intervention and time effect on spasticity{F(3,102)=0.81, p=0.489, ηp²=0.02} measured by modified Ashworth scale. While in PBS {F(3,102)=36.41, p<0.001, ηp²=0.517} significant interaction effect observed. (Figure 3)
Regarding motor function, the Friedman test indicated no overall statistically significant (p≥0.05) change in GMFCS scores over time in both the experimental and control groups and Wilcoxon signed-rank tests also confirmed no statistically significant(p≥0.05) changes between adjacent time points.
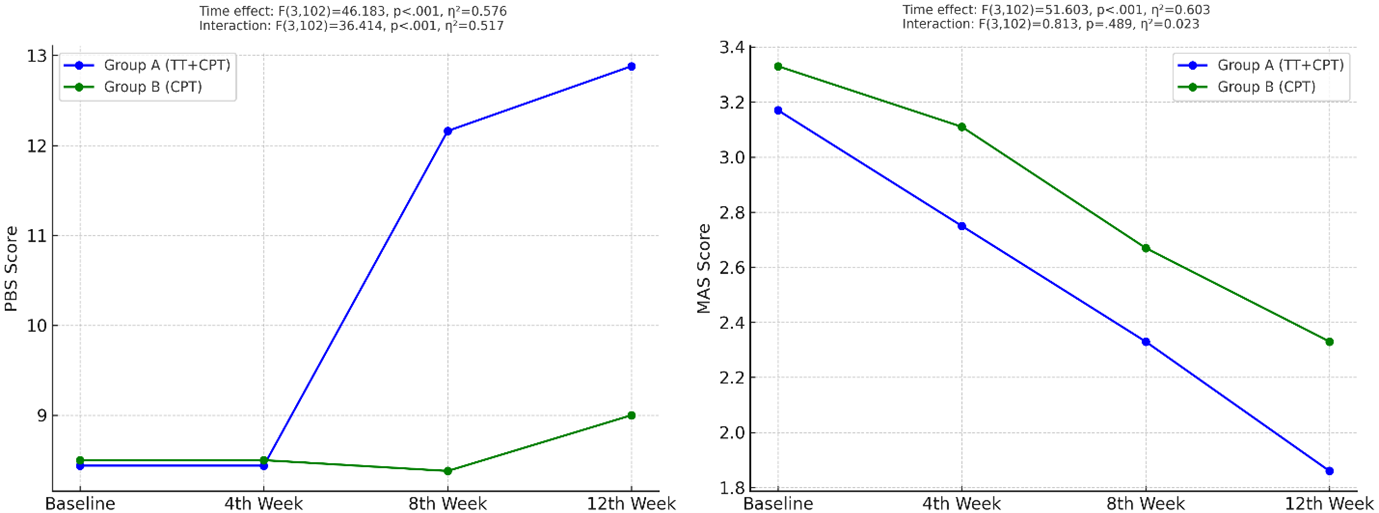
Figure 3: Interaction effect between intervention and time (PBS & Spasticity)
In group A (TT+CPT), the balance score on the Pediatric Balance Scale (PBS) and spasticity on MAS both showed significant improvements from baseline to the 12th week. The PBS score increased significantly with large effect size {8.44±5.19 to 12.88±5.68, F(3,51)=42.67, p<0.001, ɳp²=0.715}, while spasticity reduces with moderate effect size {3.17±0.71 to 1.86±1.31, F(3,51)=2.68, p<0.001, ɳp²=0.636}. Further, the Pairwise comparisons show significant improvements (p<0.05) at each time interval from baseline to 4th week, 4th to 8th week, and 8th to 12th week in both variables.
While in group B (CPT), the results indicated a significant improvement from baseline to the 12th week in both the PBS {8.50±6.28 to 19±6.38, F(3,51)=12.04, p<0.001, ɳp²=0.415} and the MAS {3.33±0.49 to 2.33±0.59, F(3,51)=18.12, p<0.001, ɳp²=0.516}. The pairwise pattern suggests that no significant improvement from baseline to the 4th week and 4th week to 8th (p≥0.05), but from the 8th to the 12th week, a statistically significant decline in PBS scores was observed (p=0.003), though the clinical relevance of this change is likely minimal. In contrast, after an initial non-significant (p=0.62) reduction from baseline to the 4th week, a significant (p=0.01) reduction in spasticity from the 8th week to the 12th week (table 2)
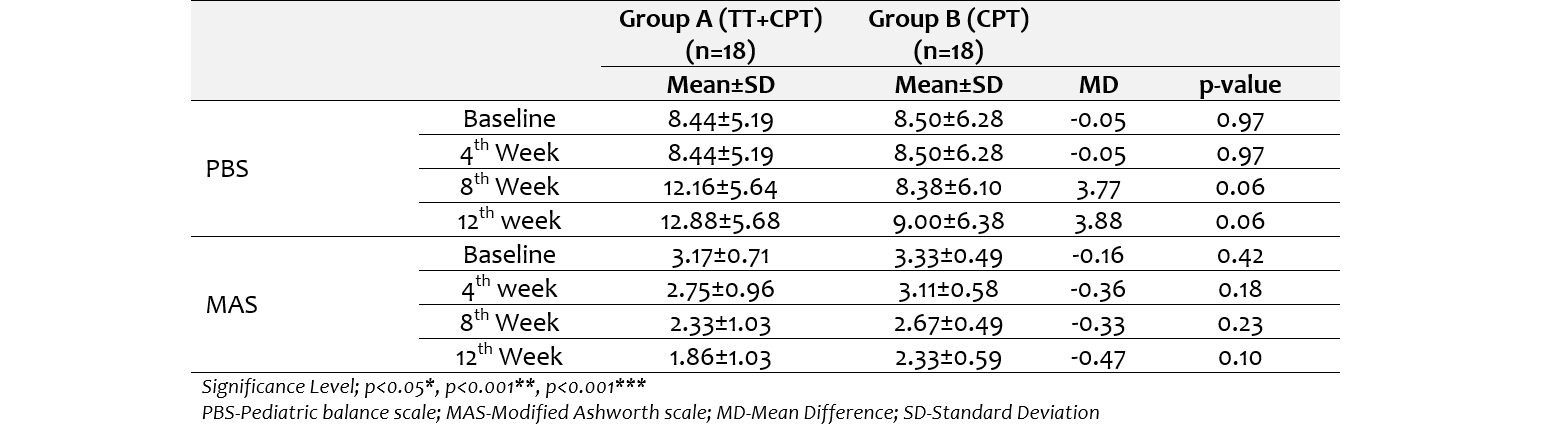
When comparing the both groups, no statistically significant differences were observed between the two groups at any time point for PBS and MAS. (table 3)
Table 2. With-in group (Main effects) changes in both groups
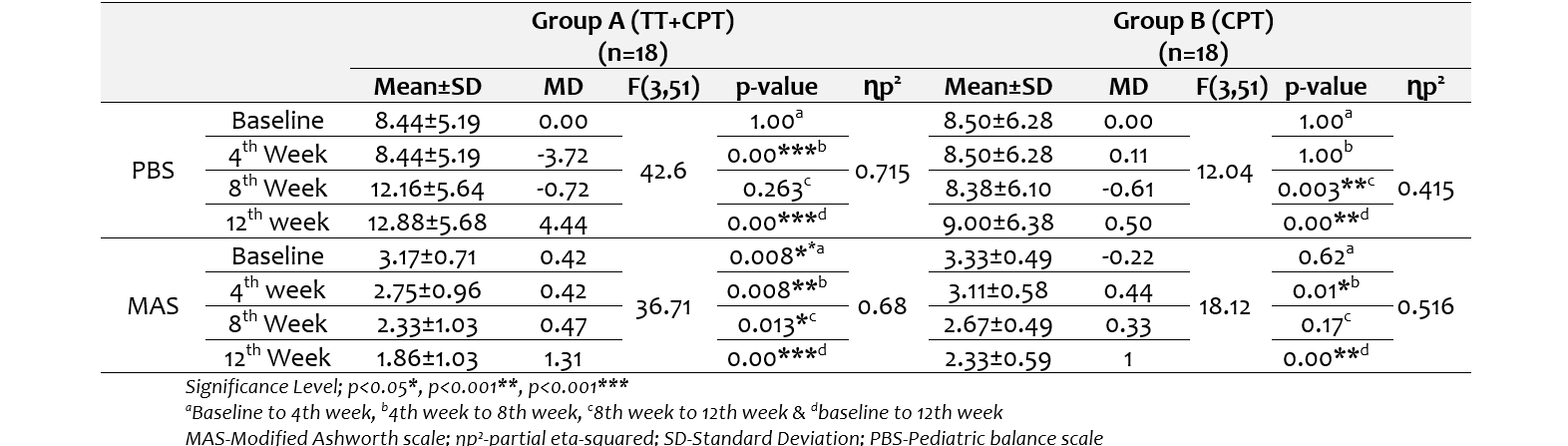
Table 3: Group comparison on PBS & MAS
DISCUSSION
The present study evaluated the effects of treadmill training along with conventional physical therapy (TT+CPT) compared to conventional physical therapy (CPT) alone on motor function, balance, and spasticity in cerebral palsy (CP).
In both groups, the most significant improvement in balance was measured on the Pediatric Balance Scale (PBS). However, the combination group (TT+CPT) demonstrated a large change with a large effect size. which suggests superior benefits of treadmill training among the study population. The progressive and task-specific nature of treadmill training may explain the enhanced gains observed in group A. Task-specific interventions that simulate gait patterns help reinforce motor learning principles and functional postural control, as proposed in dynamic systems theory [14]. The treadmill training improves functional balance by increasing postural alignment, reactive balance, and dynamic gait control[15]. Moreover, visual feedback using a mirror and verbal cues in the TT+CPT group could have motivated and facilitated proprioception and enhanced postural stability [16].
In within-group analysis, both groups showed significant improvement in balance. The TT+CPT group showed significant changes across all assessment levels from baseline to the 12th week. In contrast, the CPT group only showed significant changes from the 8th to the 12th week. This delayed response suggests that conventional therapy may take longer to produce clinically meaningful balance improvements. Treadmill-based interventions likely accelerate motor adaptation due to repetitive stepping and continuous sensory input[17, 18]. A systematic review by Sun et. al. concluded that treadmill training facilitates neural plasticity by enhancing corticospinal excitability and trunk control mechanisms [19]. Additionally, a study highlighted that gait-specific treadmill interventions promote symmetry and cadence, indirectly benefiting dynamic balance[20].
The spasticity measured by the Modified Ashworth Scale (MAS), was significantly reduced in both groups, but again, the TT+CPT group displayed earlier and greater reduction as compared to CPT alone. However, the interaction effect was non-significant, suggesting that while both interventions are effective, neither showed a statistically significant difference when compared directly. Despite this, within group, the large effect size observed in the TT+CPT group (ηp²=0.636) versus the moderate effect in the CPT group (ηp²=0.516) may show a trend towards better outcomes with treadmill training. Previous studies suggest that spasticity can be modulated through repetitive and rhythmic motion patterns, such as those provided by treadmill walking, which facilitate reciprocal inhibition and reduce hyperreflexia[21, 22].
Moreover, treadmill walking improves muscle elasticity and joint mobility, which could explain the notable reduction in MAS scores. [11].The consistent stretching and concentric-eccentric muscle activation during treadmill locomotion can reduce tonic muscle responses by engaging central pattern generators (CPGs) in the spinal cord[22, 23].
Supporting this hypothesis, Ribeirio et al found that treadmill-based gait training reduced lower limb spasticity and increased dorsiflexion range of motion[24]. Furthermore, progressively increasing intensity and duration may have enhanced descending motor control and dampened spastic neural circuits[22]. Interestingly, no significant changes were observed in motor function on Gross Motor Function Classification System (GMFCS) scores over the 12-week intervention period in either group. This outcome is not entirely surprising given that the GMFCS is a classification system rather than a responsive outcome measure. It is primarily designed to describe gross motor capacity rather than detect subtle functional changes over short-term interventions[25].
Additionally, in children older than four years, GMFCS levels are reported to be relatively stable over time, especially Hence, enhancements in dynamic skills like balance or reductions in spasticity may not necessarily translate into an immediate change in gross motor classification Instead, these gains might contribute to long-term improvements in functional independence and participation [26, 27]. A more sensitive tool such as the Gross Motor Function Measure (GMFM) may have provided better insight into motor performance improvements and responsive to changes in motor function induced by interventions in CP children[28].
Limitations Despite its strengths, including structured intervention protocols, this study has a low sample size and of short duration, as may not be sufficient to capture long-term functional changes or motor reclassification on GMFCS.GMFCS alone may not have been sensitive enough to detect functional motor improvements. Moreover, nutritional status, therapies at home, and cognitive engagement were not controlled, which may have affected the variability of responses.
CONCLUSION
The structured, progressive treadmill protocol when combined with conventional therapy, can produce clinically meaningful improvements in balance and spasticity in children with CP, even though it may not alter gross motor function classification in the short term. This combines protocol-enhanced postural stability and reduced muscle tone more effectively than conventional therapy alone, justifying its inclusion in comprehensive CP management programs. Based on current findings, future research should consider incorporating more sensitive outcome measures like GMFM, 10-meter walk test, or spatiotemporal gait parameters. Furthermore, evaluating long-term effects and maintenance achieved after stopping the treadmill training.
DECLARATIONS & STATEMENTS
Author’s Contribution
RU: substantial contributions to the conception and design of the study.
RU, HA, and OF: acquisition of data for the study.
SNG, OF and SA: interpretation of data for the study.
RU and SA: analysis of the data for the study.
RU and SA: drafted the work.
RU, HA, SNG, WF, OF, and SA: revised it critically for important intellectual content.
RU, HA, SNG, WF, OF, and SA: final approval of the version to be published and agreement to be accountable for all aspects.
of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All authors contributed to the article and approved the submitted version.
Ethical Statement
The approval obtained from the research and ethical committee of Faculty of Rehabilitation and Allied Health Sciences Islamabad (RIPHAH/FR&AHS/letter-01826) and institutional head of Helping Hand Institute of Rehabilitation Sciences, in Lower Dir Talash.
AI Use Statement
AI tools (Grammarly) were used solely for formatting and language editing. No AI was used for content generation, data analysis, or interpretation.
Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
None to declare.
Funding Sources
None to declare.
Conflicts of Interest
None to declare.
REFERENCES
- Vitrikas K, Dalton H, Breish D. Cerebral palsy: An Overview. Am Fam Physician. 2020;101(4):213-20. [PubMed]
- Saikia D, Pradhan MR. Quality of life of children with cerebral palsy: Evidence from Assam, India. Clin Epidemiol Glob Health. 2024;28:101686. [CrossRef] [PubMed]
- Khan SA, Talat S, Malik MI. Risk factors, types, and neuroimaging findings in children with cerebral palsy. Pak J Med Sci. 2022;38(7):1738-42. [CrossRef] [PubMed]
- Rafique A. The burden of cerebral palsy in Pakistan: an insight into demographic and preventive planÇukurova med. j. 2022;47(4):1759-60. [CrossRef]
- Wilson JL, Aravamuthan BR, O’Malley JA. Cerebral palsy. In: kamat dm, sivaswamy l, editors. Symptom-based approach to pediatric neurology. Cham: Springer International Publishing; 2023. p. 541-64. [CrossRef]
- Smith SE, Gannotti M, Hurvitz EA, Jensen FE, Krach LE, Kruer MC, et al. Adults with cerebral palsy require ongoing neurologic care: A Systematic Review. Ann Neurol. 2021;89(5):860-71. [CrossRef] [PubMed]
- Rapson R, Latour JM, Marsden J, Hughes H, Carter B. Defining usual physiotherapy care in ambulant children with cerebral palsy in the United Kingdom: A mixed methods consensus study. Child Care Health Dev. 2022;48(5):708-23. [CrossRef] [PubMed]
- Bonanno M, Militi A, La Fauci Belponer F, De Luca R, Leonetti D, Quartarone A, et al. Rehabilitation of gait and balance in cerebral palsy: a scoping review on the use of robotics with biomechanical implications. J Clin Med. 2023;12(9). [CrossRef] [PubMed]
- Alotaibi A, Ibrahim A, Ahmed R, Abualait T. Effectiveness of partial body weight-supported treadmill training on various outcomes in different contexts among children and adolescents with cerebral palsy: A Systematic Review and Meta-Analysis. Children (Basel). 2023;11(1). [CrossRef] [PubMed]
- Gunawan PI, Lusiana L, Noviandi R, Samosir SM. Combination of standard physical therapy with partial body weight supported treadmill training in children with spastic diplegic cerebral palsy. International journal of health sciences. 2022;6(S9):1866-77. [CrossRef]
- Han YG, Yun CK. Effectiveness of treadmill training on gait function in children with cerebral palsy: meta-analysis. J Exerc Rehabil. 2020;16(1):10-19. [CrossRef] [PubMed]
- Bishnoi A, Lee R, Hu Y, Mahoney JR, Hernandez ME. Effect of treadmill training interventions on spatiotemporal gait parameters in older adults with neurological disorders: systematic review and meta-analysis of randomized controlled trials. Int J Environ Res Public Health. 2022;19(5). [CrossRef] [PubMed]
- Chin EM, Gwynn HE, Robinson S, Hoon AH, Jr. Principles of medical and surgical treatment of cerebral palsy. Neurol Clin. 2020;38(2):397-416. [CrossRef] [PubMed]
- Krishnan C, Dharia AK, Augenstein TE, Washabaugh EP, Reid CE, Brown SR, et al. Learning new gait patterns is enhanced by specificity of training rather than progression of task difficulty. J. Biomech. 2019;88:33-7. [CrossRef]
- Rieger MM, Papegaaij S, Steenbrink F, van Dieën JH, Pijnappels M. Effects of perturbation-based treadmill training on balance performance, daily life gait, and falls in older adults: react randomized controlled trial. Phys Ther. 2024;104(1). [CrossRef] [PubMed]
- Kasuga S, Crevecoeur F, Cross KP, Balalaie P, Scott SH. Integration of proprioceptive and visual feedback during online control of reaching. J Neurophysiol. 2022;127(2):354-72. [CrossRef] [PubMed]
- Sharma P, Gupta M, Kalra R. Recent advancements in interventions for cerebral palsy – A review. Journal of Neurorestoratology. 2023;11(3):100071. [CrossRef]
- Winter L, Huang Q, Sertic JVL, Konczak J. The effectiveness of proprioceptive training for improving motor performance and motor dysfunction: a systematic review. Frontiers in Rehabilitation Sciences. 2022;Volume 3 - 2022. [CrossRef]
- Sun Y, Hurd CL, Barnes MM, Yang JF. Neural plasticity in spinal and corticospinal pathways induced by balance training in neurologically intact adults: a systematic review. Front Hum Neurosci. 2022;16:921490. [CrossRef] [PubMed]
- Zhang M, Liang Z, Li Y, Meng J, Jiang X, Xu B, et al. The effect of balance and gait training on specific balance abilities of survivors with stroke: a systematic review and network meta-analysis. Front Neurol. 2023;14:1234017. [CrossRef]
- Thompson AK, Wolpaw JR. H-reflex conditioning during locomotion in people with spinal cord injury. J Physiol. 2021;599(9):2453-69. [CrossRef] [PubMed]
- Ganguly J, Kulshreshtha D, Almotiri M, Jog M. Muscle tone physiology and abnormalities. Toxins (Basel). 2021;13(4). [CrossRef] [PubMed]
- Merlet AN, Harnie J, Frigon A. Inhibition and facilitation of the spinal locomotor central pattern generator and reflex circuits by somatosensory feedback from the lumbar and perineal regions after spinal cord injury. Frontiers in Neuroscience. 2021;Volume 15 - 2021. [CrossRef] [PubMed]
- Ribeiro TS, Silva E, Silva IAP, Costa MFP, Cavalcanti FAC, Lindquist AR. Effects of treadmill training with load addition on non-paretic lower limb on gait parameters after stroke: A randomized controlled clinical trial. Gait Posture. 2017;54:229-35. [CrossRef] [PubMed]
- Towns M, Rosenbaum P, Palisano R, Wright FV. Should the gross motor function classification system be used for children who do not have cerebral palsy? Dev Med Child Neurol. 2018;60(2):147-54. [CrossRef] [PubMed]
- Park EY. Stability of the gross motor function classification system in children with cerebral palsy for two years. BMC Neurol. 2020;20(1):172. [CrossRef] [PubMed]
- Wizinsky AM, Donawerth M, Badgley M, Hemphill-Morytko B, Laughlin E, Rogan M. Stability of the gross motor function classification system in children with cerebral palsy in the two to four year age band. J Pediatr Rehabil Med. 2023;16(2):321-9. [CrossRef]
- Choi JY. Motor function measurement in children: gross motor function measure (gmfm). Ann Rehabil Med. 2024;48(5):301-4. [CrossRef] [PubMed]
Submitted
Accepted
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Rizwan Ullah, Hifza Arif, Sajjal Naeem Gul, Waqas Farooq, Osama Faisal, Sheeraz Ali

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors retain copyright and grant the journal right of first publication and allows others to share the work with an acknowledgment of the work’s authorship and initial publication in this journal. No use, distribution or reproduction is permitted which does not comply with these terms.